An international research team, led by Tibor Harkany and Robert Schnell at Karolinska Institutet and MedUni Vienna’s Center for Brain Research, set out to find ways of influencing hormone release to reduce stress reactions in post-traumatic stress disorder. They came across secretagogin, which regulates the release of some of the most fundamental and indispensable neurotransmitters and hormones.
In this study, the role of secretagogin was identified through its interactions with two core proteins of the release (“SNARE”) machinery. The authors have identified a new partner for secretagogin, which is a calcium-sensor protein that is particularly abundant at synapses. This partner, syntaxin-4, is required for vesicles to move towards the release site. The other, earlier established partner is snap-25. Both proteins are integral to the SNARE complex. Secretagogin binds snap-25 with an affinity higher than syntaxin-4.
“This is important because, based on chemical principles, secretagogin can “hand over” syntaxin-4-tagged cargo vesicles for release once snap-25 is engaged”, says Robert Schnell, Associate Professor at the Department of Neuroscience at Karolinska Institutet.
The data are compatible with earlier studies, which show that the lack of secretagogin specifically impairs second phase release, which involves vesicle trafficking to release sites. This principle, and secretagogin expression, is relevant to many excitable neuroendocrine tissues, e.g., the hypothalamus and pancreas.
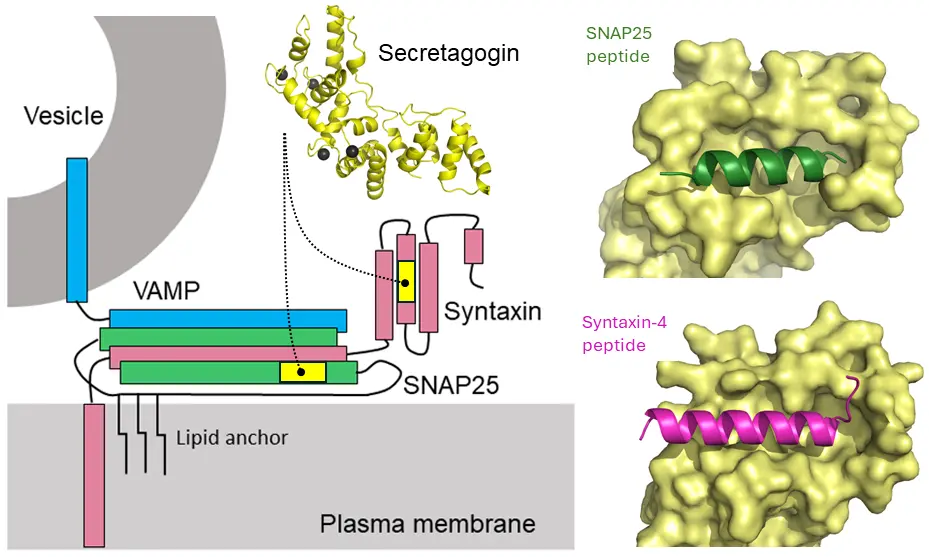
Image: Robert Schnell
The research team used protein biochemistry, structural biology, and cellular models to perform the study. Protein complexes were also crystallized. The structures of three protein complexes were determined by X-ray crystallography.
The next step
The new information supports the development of inhibitors blocking the secretagogin-SNARE interactions. There is an entirely new principle for drug development, with small molecules potentially affecting the rate of hormone release in a cell-type specific manner.
The study was mainly funded via an ERC Proof of Concept Grant and the Novo Nordisk Foundation.
Publication
Szodorai E, Hevesi Z, Wagner L, Hökfelt TGM, Harkany T, Schnell R
Proc Natl Acad Sci U S A 2024 Apr;121(16):e2309211121


.png)





