A team of researchers at Tohoku University has reported on a novel function of vitamin K, which is generally known for its importance in blood clotting. The researchers discovered that the fully reduced form of vitamin K acts as an antioxidant efficiently inhibiting ferroptotic cell death. Ferroptosis is a natural form of cell death that is characterized by extensive lipid peroxidation in cellular membranes. In addition, the team identified FSP1 as the warfarin-insensitive enzyme reducing vitamin K, the identity of which had been postulated but remained unknown for more than half a century. Recently, ferroptosis has been implicated as a driver of Alzheimer’s disease and acute organ injuries among many other diseases. The findings suggest that vitamin K treatment might be a new powerful strategy to ameliorate these ferroptosis-related diseases.
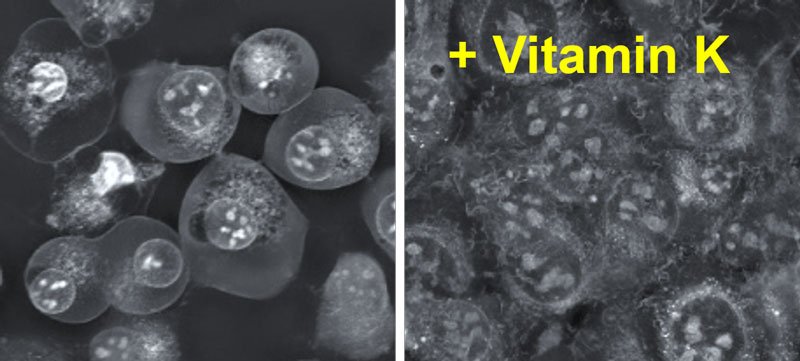
Vitamin K is a potent ferroptosis suppressor
Since ferroptosis prevention is considered a highly promising approach for the therapy of many degenerative diseases, new mechanisms and compounds regulating ferroptosis are extensively being explored. To identify these new molecules, a team of researchers led by Dr Eikan Mishima (Tohoku University) and Dr Marcus Conrad (Helmholtz Munich), systematically studied several naturally occurring vitamins, as well as their derivatives. “Surprisingly, we identified that vitamin K, including phylloquinone (vitamin K1) and menaquinone-4 (vitamin K2), are able to efficiently rescue cells and tissues from undergoing ferroptosis” Dr Mishima explained.
Unravelling the long-sought-after vitamin K reducing enzyme FSP1
In 2019 a team of researchers, led by Dr Conrad, identified an enzyme as a novel and strong inhibitor of ferroptosis: ferroptosis suppressor protein-1, short FSP1. The current research team has now found that the fully reduced form of vitamin K (i.e., vitamin K hydroquinone) acts as a strong lipophilic antioxidant and prevents ferroptosis by trapping oxygen radicals in lipid bilayers. In addition, they identified that FSP1 is the enzyme that efficiently reduces vitamin K to vitamin K hydroquinone, thereby driving a novel non-canonical vitamin K cycle. Since vitamin K is critically involved in blood clotting processes, the team additionally showed that FSP1 is responsible for the vitamin K-reduction pathway insensitive against warfarin, which is one of the most prescribed anticoagulants.
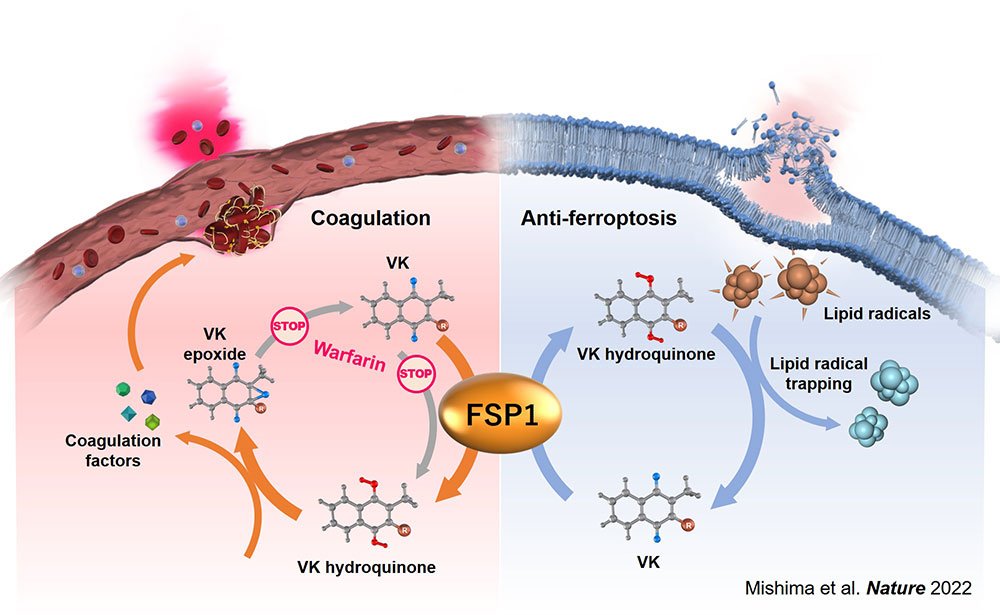
(Left) FSP1 also functions as the warfarin-resistant vitamin K reduction pathway overcoming warfarin poisoning. ©Tohoku University
Breakthrough in understanding vitamin K metabolism
Unravelling the identity of this enzyme solved the last riddle of vitamin K metabolism in blood clotting and elucidated the molecular mechanism of why vitamin K constitutes the antidote for warfarin overdosing. Dr Mishima and Dr Conrad have indicated that “our results have the potential to connect the two worlds of ferroptosis research and vitamin K biology. They will serve as a stepping stone for the development of novel therapeutic strategies for diseases where ferroptosis has been implicated.” In addition, since ferroptosis most likely constitutes one of the oldest types of cell death, the researchers hypothesize that vitamin K might be one of the most primitive types of naturally occurring antioxidants. “Thus, new aspects of the role of vitamin K throughout the evolution of life are expected to be unveiled.”
- Publication Details:
Title: A non-canonical vitamin K cycle is a potent ferroptosis suppressor
Authors: Eikan Mishima, Junya Ito, Zijun Wu, Toshitaka Nakamura, Adam Wahida, Sebastian Doll, Wulf Tonnus, Palina Nepachalovich, Elke Eggenhofer, Maceler Aldrovandi, Bernhard Henkelmann, Ken-ichi Yamada, Jonas Wanninger, Omkar Zilka, Emiko Sato, Regina Feederle, Daniela Hass, Adriano Maida, André Santos Dias Mourão, Andreas Linkermann, Edward K. Geissler, Kiyotaka Nakagawa, Takaaki Abe, Maria Fedorova, Bettina Proneth, Derek A. Pratt & Marcus Conrad
Journal: Nature








