Flat batteries could be a thing of the past thanks to lithium-ion battery nanotechnology developed by The University of Queensland.
The technology more than doubles the lifespan of highly sought-after high-voltage Li-ion batteries, which achieve higher energy density but last only several hundred cycles.
Professor Lianzhou Wang and his team from the School of Chemical Engineering and Australian Institute for Bioengineering and Nanotechnology (AIBN) have demonstrated a battery which remains stable for more than 1000 charge/discharge cycles.
“Our process will increase the life-span of batteries in many things from smart phones and laptops, to power tools and electric vehicles,” Professor Wang said.
“We’ve designed a uniquely-grown atomic-thin functional layer on the surface of a high-voltage cathode, which is the source of lithium ions and a critical aspect that limits the cycle life in a battery.
“Corrosion in one form or another is the reason that batteries degrade over time.
“This new approach features a minimal protective coating at a scalable process, paving the way for the deployment of these abundant high-voltage materials for next generation high energy batteries.”
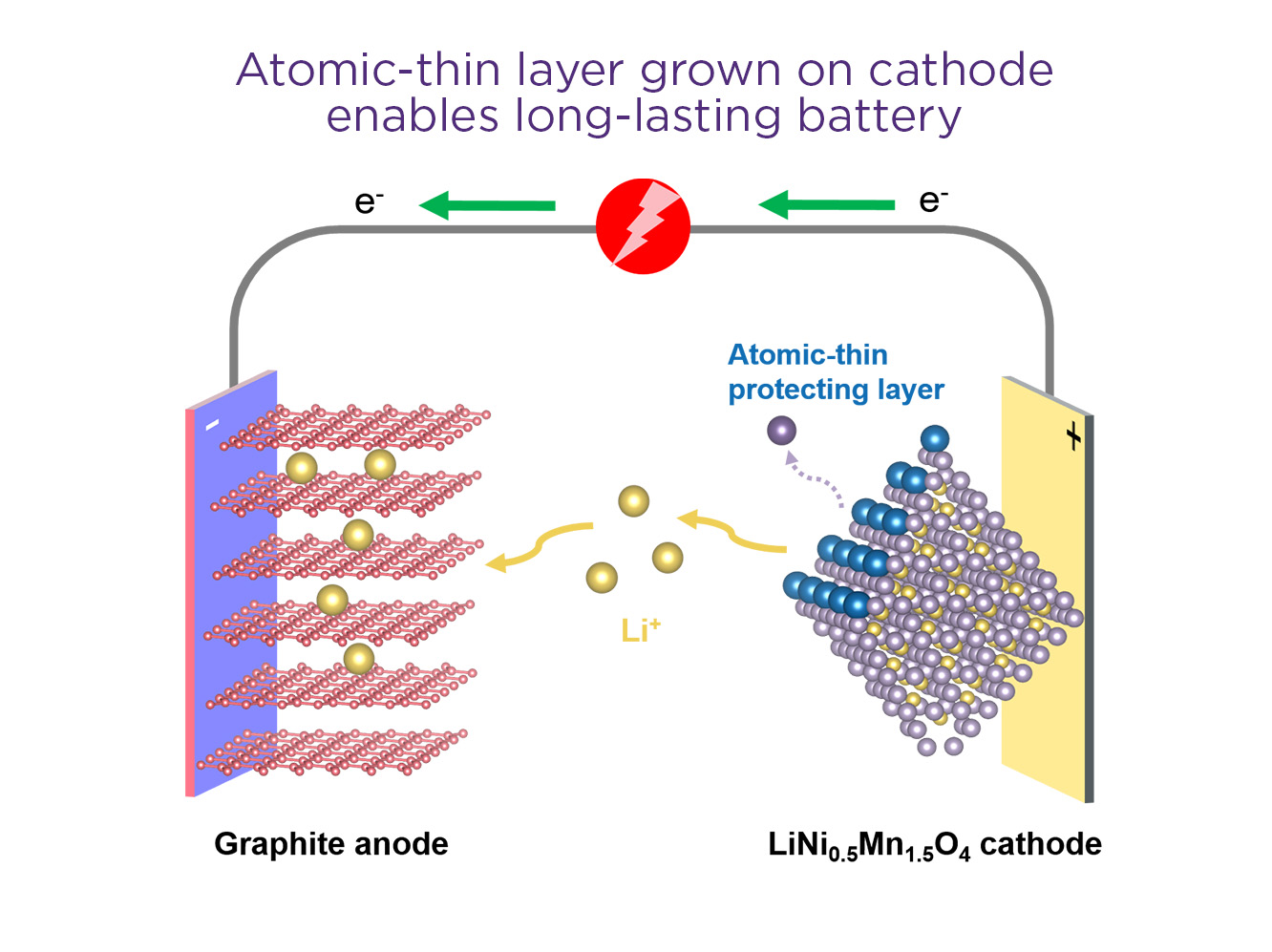 Professor Wang said with industry under increasing pressure to decarbonise, the development of lithium-ion batteries of lower cost, higher energy density and longer cycle life was vitally important.
Professor Wang said with industry under increasing pressure to decarbonise, the development of lithium-ion batteries of lower cost, higher energy density and longer cycle life was vitally important.
“We’re confident the nanotechnology will have widespread applications across industry, including in consumer electronics, electric vehicles and the energy storage sector,” he said.
Dr Rosalind Gummow, Technical Specialist from Brisbane-based R&D company VSPC Pty Ltd, which is focused on developing and commercialising advanced cathode materials for lithium-ion batteries, welcomed this development.
“New methods like the use of epitaxial surface layers to improve the cycling efficiency and cycle life of high-voltage cathodes are vital in the quest to improve the energy density of Li-ion batteries,” Dr Gummow said.
“The methods developed here also have potential to stabilise other cathode materials that degrade rapidly with cycling.”
This latest research was published in Nature Communications.








