New research from the University of Washington and Northwestern University shows that immune cells ‘count’ how many of them have gathered to determine how much the immune system should react.Northwestern University
Many people consult their friends and neighbors before making a big decision. It turns out that cells also consult their neighbors – in the human body.
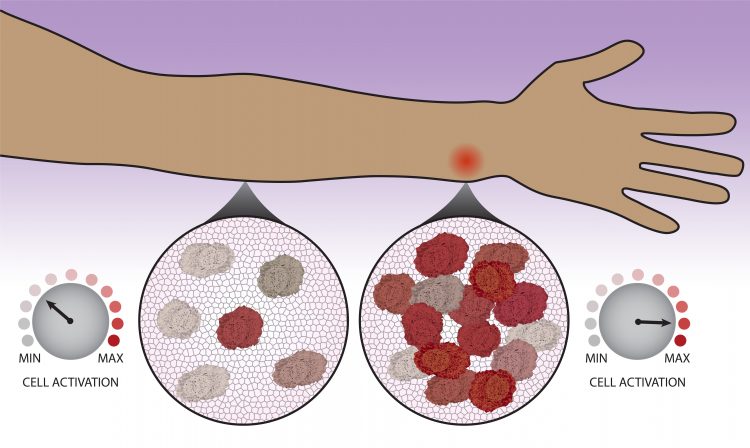
Scientists and physicians have long known that immune cells migrate to the site of an infection, which individuals experience as inflammation – swelling, redness and pain. Now, researchers at the University of Washington and Northwestern University have uncovered evidence that this gathering is not just a consequence of immune activation. Immune cells count their neighbors before deciding whether or not the immune system should kick into high gear.
Understanding how to influence inflammation and activate an immune response could lead to new therapies to treat chronic autoimmune diseases or to mobilize the immune system to help fight cancer.
“This is a previously unrecognized aspect of immune function,” said Joshua Leonard, a professor of chemical and biological engineering at Northwestern. “The cells make a coordinated decision. They don’t uniformly activate but instead collectively decide how many cells will activate, so that together, the system can fend off a threat without dangerously overreacting.”
“A key part of this work relied on the development of new computational models to interpret our experiments and elucidate how cells perform calculations to make coherent decisions,” said Neda Bagheri, an assistant professor of biology and chemical engineering at the UW.
Bagheri and Leonard co-led the study, which was published Feb. 13 in Nature Communications.
The body’s immune system works constantly to maintain a delicate balance. When a threat is introduced, the system needs to respond strongly enough to fight off infection or disease, but not so strongly that it causes harm.
“When it comes to immune responses, it’s the difference between life and death,” Leonard said. “If your body over-responds to a bacterial infection, then you could die from septic shock. If your body doesn’t respond enough, then you could die from rampant infection. Staying healthy requires the body to strike a balance between these extremes.”
Bagheri, Leonard and their teams wanted to better understand how the immune system makes these types of decisions.
“It’s especially interesting because the immune system is decentralized,” said lead author Joseph Muldoon, a graduate student at Northwestern University who is co-advised by Bagheri and Leonard. “Immune cells are individual agents that need to work together, and nature has come up with a solution for how they can get on the same page. Cells arrive at different activation states, but in such a way that, on the whole, the population response is calibrated.”
To explore this phenomenon, the researchers examined macrophages, a type of immune cell that is part of the first line of defense for combatting infection and disease. They observed how macrophages responded to a chemical produced by bacteria – a red flag that alerts the body to the presence of infection – using techniques that enabled the researchers to watch individual cells’ responses over time. They then used computational models to help interpret and explain these observations.
Immune cells were ‘counting’ how many of them had gathered to determine how much the system should react.
“Over time, the cells observe their surroundings to get a sense of their neighbors,” Muldoon said. “Each cell becomes poised to respond as a high activator or not. Now that we know there’s this additional layer controlling the immune system, it opens up a whole avenue to study whether there are new targets for immunomodulation.”
The researchers believe this information could be used to develop improved cancer immunotherapies or treatments for autoimmune diseases, help design better drugs and guide the engineering of advanced cell-based therapies.
Co-author on the paper is Yishan Chuang, who conducted this work as a graduate student at Northwestern. The research was funded by the National Institutes of Health and Northwestern University.







